Acid Fuchsin C.I. 42685
Properties
UV-VIS range: 542-546
Min. Dye Content: 60%
Molecular Formula: C20H17N3Na2O9S3 or C20H17CaN3O9S3
Formula Weight: 585.55 or 579.65
Appearance: Green Crystaline Powder
Dye Class: Aminotriarylmethane
Biological Applications: Widely used plasma stain, which also has many other uses including staining connective tissue, mitochondria, and unaborted pollen grains.
Commercial Applications: Textile Dye, dyeing of crepe paper, soap, and photographic film.
Altmann’s Stain for Mitochondria
Preparation of Stain Solutions
1% Aqueous Methyl Green
50 ml of Distilled Water
500 mg of Methyl Green
Let the mixture sit overnight to dissolve completely
Aniline Acid Fuchsin
Add a few drops of Aniline to 12ml of distilled water and saturate
Shake solution and filter.
Use 10ml of the filtrate and add 1g of Acid Fuchsin
Dissolve completely, filter and pour into a dropper bottle.
Tissue Preparation: Fixation – Formalin Dichromate Control – Kidney Tissue (Use Mullers or Regaud’s fluid as a pretreatment to embedding after formalin fixation and processing.)
Staining Procedure
1. Deparafinize sections through xylene.
2. Hydrate tissues through graded ethanol.
3. Rinse sections in distilled water.
4. While holding the slide flat with forceps, add a few drops of aniline acid fuchsin to the section.
5. Heat the section over an open flame until it begins to steam. (be careful not to overheat the section).
6. Let the slide cool and continue staining for 5 minutes.
7. Drain off the dye and rinse the slide in distilled water for 3 minutes.
8. Place the slide into the Methyl Green solution for 30sec to 10minutes. Look for differentiation, you may rinse the slide in 95%ethanol and examine under the microscope. (The methyl green solution will remove or replace some of the red staining.)
9. Differentiate the Methyl Green staining in 95% ethanol.
10. Run down through graded alcohols, xylene and mount.
Results
Mitochondira – red
Nuclei – green
Granules – red
Van Gieson's Stain
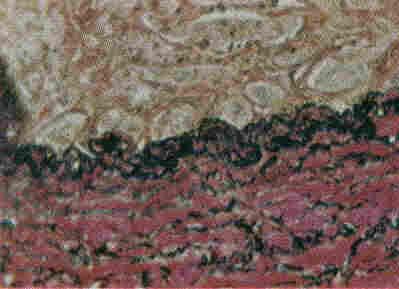 Preparation of Stain Solutions
Preparation of Stain Solutions
1% aqueous acid fuchsin solution
Saturated aqueous picric acid solution
Weigert's iron Hematoxylin
Stock Solution A
Hematoxylin 1gm
95% ETOH 100 ml
Stock Solution B
29% aquesous ferric chloride 4ml
Distilled Water 95ml
Hydrochloric acid 1ml
Working Solution - Equal parts of stock solutions A and B
Van Gieson's Staining Solution
1% aquesous acid fuchsin 5ml
Saturated aqueous Picric acid 50ml
Distilled Water 50ml
Aniline Acid Fuchsin
Add a few drops of Aniline to 12ml of distilled water and saturate
Shake solution and filter.
Use 10ml of the filtrate and add 1g of Acid Fuchsin
Dissolve completely, filter and pour into a dropper bottle.
Staining Procedure
- Deparafinize sections through xylene.
- Hydrate tissues through graded ethanol.
- Rinse sections in distilled water.
- Place sections in Weigert's iron hematoxylin solution for 10-20 minutes.
- Rinse sections in running tap water for 10 minutes.
- Place sections in Van Gieson's stain for 5 minutes.
- Place slides in 95% ethanol and dehydrate through 100% ethanol..
- Clear through xylene and mount with a synthetic resin medium.Results Collagen - red Smooth and striated muscle - yellow Nuclei - blue-black
REFERENCES:
Lillie, R.D. 1977. H.J. Conn's Biological Stains, 9th edition. Williams & Wilkins, Baltimore. pp. 588-589,285-286.
Sheenan, D.C., B.B. Hrapchak. Theory and practice of histotechnology, 2nd edition. Battelle Press, Columbus. pp. 189.
Green, F.J. 1990. The Sigma-Aldrich Handbook of Stains, Dyes and Indicators. Aldrich Chemical Comnpany, Milwaukee. pp.19-21.
Conn, H.J, M.A. Darrow, V.M. Emmel 1960. Staining Procedures, 5th edition. Williams & Wilkins, Baltimore. pp.165-166.